Red-Edge Excitation Spectroscopy of Protein Fluorescence with the FS5 Spectrofluorometer
The study of Protein Fluorescence is an important area of research in biology and biophysics. Red-edge fluorescence spectroscopy of polar fluorescence probes is a powerful method to study the structure and dynamics of phospholipid membranes, micelles and proteins. This method takes advantage of the possibility to selectively excite at the red edge of the absorption band, a small fraction of fluorescence probes with red-shifted emission spectrum. Position of the emission maximum of these red-shifted fluorescence probes yields information about the polarity and dynamics of a protein’s microenvironment. It is a steady-state method and does not require expensive instrumentation. With the FS5 Spectrofluorometer it is possible to work with micro-cuvettes and still maintain high sensitivity of fluorescence detection. This was a necessity with this method as it requires very small amounts of protein. The FS5 is a powerful instrument and is ideally suited for this type of application.
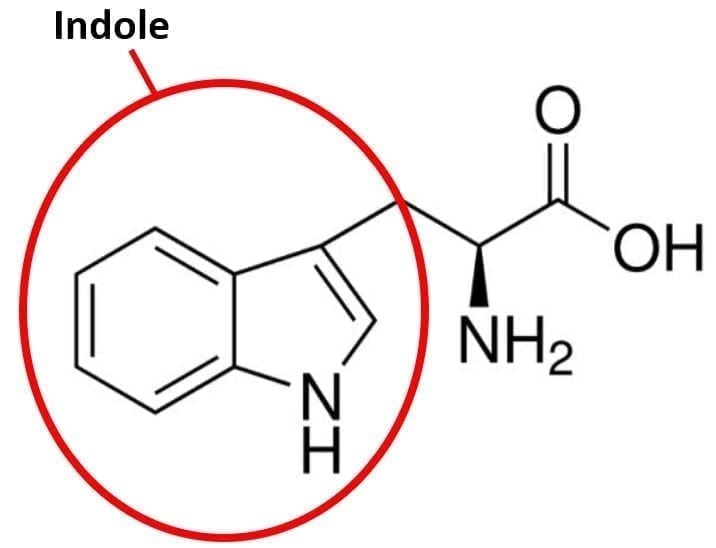
Figure 1: Tryptophan
Tryptophan fluorescence has found a broad scope of applications in life sciences. Tryptophan (amino acid) is a derivative of indole, which is a polar dye molecule with a large permanent dipole moment in the ground (2.2 D) and excited (5.7 D) states. This makes tryptophan fluorescence sensitive to polarity and viscosity. In proteins, each tryptophan site chain is situated in a cavity (pocket) with a unique polarity and molecular dynamics. The tryptophan fluorescence pattern of a protein, containing only one tryptophan residue, shows similar properties as a viscous polar dye solution. This can be rationalised by a dynamic model of inhomogeneous broadening of electronic spectra, which accounts for a spectral relaxation of the solvated molecule in the excited state.
Download the full Protein Fluorescence Application Note
Red-Edge Excitation Spectroscopy of Protein Fluorescence with the FS5 Spectrofluorometer
FS5 Spectrufluorometer
For further information on the FS5 Spectrofluorometer, why not contact one of our sales team today at: sales@edinst.com.
Sign up for our Application and Technical Notes
If you have enjoyed reading our Application Note, why not sign-up to our infrequent newsletter via our red Sign-up button below.