Understanding the Light-Harvesting Photosystem II; Utilising Transient Absorption to Study a Molecular Triad’s Photoinduced Electron Transfer Reactions
Introduction
The ability of plants to live, thrive and survive on sunlight provides unsurpassed motivation to understand and mimic this process for solar energy production. Photosystem II has long been studied and admired by scientists eager to harness its energetic potential; absorbed sunlight produces a cascade of uphill, proton coupled electron transfer (PCET) events that funnel energy for plants to utilise to synthesise food. The Edinburgh Instruments LP980 Transient Absorption (Flash Photolysis) Spectrometer is the world’s only commercially available system to feature dual detector options for direct kinetic and spectral measurements required to measure such energy and electron transfer events, and is ideally suited for solar energy research.

Figure 1: The Edinburgh Instruments LP980 Spectrometer.
Research
Andrea Pannwitz and Professor Oliver S. Wenger, who perform inspiring photochemical research into electron and energy transfer mechanisms at the University at Basel in Switzerland, utilised transient absorption to study, for the first time, a photo-induced long-range electron transfer coupled with acceptor protonation and donor deprotonation in a molecular triad designed to mimic Photosystem II (PSII).
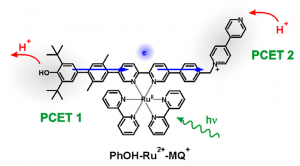
Figure 2: The studied molecular triad designed to mimic Photosystem II (PSII) showing multiple proton coupled electron transfers (PCET) and long-range electron transfer.
These researchers methodically designed a molecular triad to behave similarly to PSII; a phenol donor (tyrosine Z mimic), linked to a Ru(II) sensitiser (P680 mimic), and additionally linked to a monoquat acceptor (plastoquinone B mimic). Photoexcitation of the Ru(II) sensitizer initiates the process much like P680 in PSII, and generates a metal-to-ligand charge transfer (MLCT) excited state (Figure 2). A PCET event then takes place on the phenol donor (PCET 1), generating a phenoxy radical, and undergoing long range electron transfer to the monoquat, where a subsequent PCET event takes place (PCET 2). By varying the solvent, and utilising transient absorption spectra from the ICCD and PMT detectors to study the photogenerated radical spectra and lifetime, respectively, and comparing these to the spectroelectrochemistry spectra to prove the radical species present in the photogenerated transient spectra, the researchers were able to prove multi-step PCET and long-range electron transfer events (Figure 3).
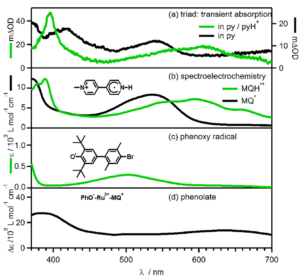
Figure 3: (A) Photogenerated transient absorption spectra of the molecular triad, (B) spectroelectrochemistry of the monoquat, (C) absorption of the phenoxy radical, and (D) difference spectra showing the absorption of phenolate.
Conclusion
This study highlights how designing unique molecular architecture can give insights into nature’s ability to harvest and store solar energy efficiently and avoid the build-up of charge; these proton coupled electron transfer processes can be directly studied by the Edinburgh Instruments LP980 Transient Absorption Spectrometer.
Link to Article: https://pubs.acs.org/doi/abs/10.1021/jacs.7b08761
Figures reprinted with permission from J. Am. Chem. Soc. 2017, 139, 13308-13311.









