Time-resolved Spectroscopy of Phosphorescent Oxygen Sensors in a Relevant in vitro Environment for Biomedical Applications
Kayla F. Presleya,b, Jack T. Lya,b, James R. Deneaulta,b, Matthew J. Daltona, Tod A. Grusenmeyera
a Air Force Research Laboratory, Materials and Manufacturing Directorate, 2179 12th St, Wright-Patterson AFB, OH 45433, USA
b UES, Inc., 4401 Dayton-Xenia Road, Dayton, OH 45432, USA
Optical oxygen sensors generally function based on incorporation of a phosphorescent molecule, such as a transition metal polypyridyl complex or heavy metal porphyrin, into a polymer matrix (e.g., thin film, electrospun fibers, etc.). The long-lived luminescence from the incorporated molecule is dynamically quenched in the presence of oxygen. Considering the phosphorescence quantum yield in the presence or absence of oxygen (Eq. 1) yields the Stern-Volmer relationship (Eq. 2) which governs this dynamic quenching process. KSV represents the Stern-Volmer quenching constant and indicates the sensitivity of a given oxygen sensor to dissolved oxygen in the surrounding environment. This parameter is equal to the product of the lifetime in the absence of oxygen, Ƭ0, and the bimolecular quenching constant, kq (Eq. 3).
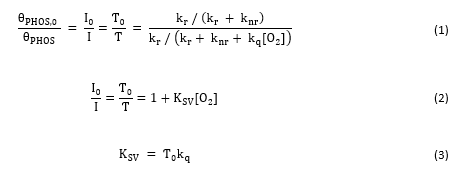
Optical oxygen sensors can be used for both gaseous and dissolved oxygen sensing and have a wide variety of applications, such as food packaging, pressure sensitive paints, assessing oxygen levels in cell culture, monitoring tissue oxygenation, etc. Many factors, such as the application temperature(s), the relevant range of oxygen concentrations, and the state of sensor hydration, can vary widely among different applications, and such factors can have a significant impact on sensor behavior. Therefore, if a sensor is developed for a particular application, performing initial photophysical characterization under relevant conditions is critical.
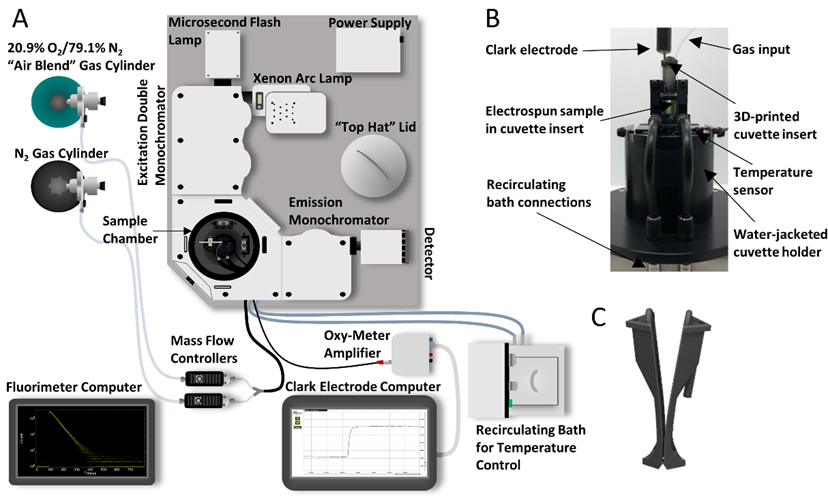
FLS1000 setup for relevant in vitro characterization of candidate tissue oxygenation sensors: (A) FLS1000 setup schematic (B) Water-jacketed cuvette holder with cuvette insert holding a candidate sensor (C) Cuvette insert design. (Reprinted from Sensors and Actuators B: Chemical, 329, Presley, K.F., Reinsch, B.M., Cybyk, D.B., Ly, J.T., Schweller, R.M., Dalton, M.J., Lannutti, J.J. and Grusenmeyer, T.A., Oxygen sensing performance of biodegradable electrospun nanofibers: Influence of fiber composition and core-shell geometry, 129191, Copyright (2021), with permission from Elsevier. (https://www.sciencedirect.com/journal/sensors-and-actuators-b-chemical)
This customer written application note details how researchers from the Air Force Research Laboratory have outfitted an Edinburgh Instruments FLS1000 Photoluminescence Spectrometer to assess candidate dissolved oxygen sensors. Using the FLS1000, the researchers assessed the performance of different oxygen sensors in a relevant in vitro environment in order to evaluate their potential for use as an implantable sensor that could be used to continuously monitor tissue oxygenation in vivo. The sensor performance was characterized via changes in the phosphorescence lifetime which was measured using Multi-Channel Scaling (MCS) and a microsecond xenon flash lamp.
Discover the FLS1000 Photoluminescence Spectrometer
If you are working with Optical Oxygen Sensors and would like to know how the FLS1000 Spectrometer can help you with your research, please contact us.
Keep in Touch
If you have enjoyed reading this application note, why not sign-up to our monthly eNewsletter via the button below or follow us on your favourite social media channel in the footer below to keep up to date with our latest news and research.









