Raman Spectroscopy for Polymorph Identification in Pharmaceuticals
Key Points
- Polymorphism in pharmaceuticals is the formation of different crystallographic forms of the same drug compound.
- Polymorphs are a challenge in manufacturing because they can reduce the efficacy of treatments.
- The high spectral resolution of Raman spectroscopy means it can detect subtle spectral differences between different polymorphs of the same drug.
Introduction
In the pharmaceutical industry, drug manufacturing techniques are highly complex and have a meaningful effect on the safety and efficacy of the final product. One phenomenon that can arise during this process is polymorphism, which is the formation of different crystallographic forms of the same drug compound. The identification of polymorphic forms is essential for several reasons:
- Different polymorphs in a formulation can reduce its chemical and physical stability, negatively impacting the product’s shelf life.
- Different particle morphologies into formulations can cause processing issues during manufacturing that need to be accounted for.
- Polymorphs can have different solubilities and dissolution rates upon being administered to the patient, properties that are closely linked to bioavailability.1
With knowledge and control of the crystal structure multiplicity within a formulation, the effectiveness of the active ingredient can be optimised.
The most well-known and established method for the identification of polymorphs is X-ray diffraction (XRD) analysis. It is commonly used for analysing crystals because of the wealth of information it provides about the structure and composition of the sample. However, its drawbacks include the inability to measure amorphous substances and a large laboratory footprint.
Raman spectroscopy is a technique that offers detailed chemical information but can also overcome the disadvantages mentioned above associated with XRD. Raman is an excellent technique for identifying polymorphs, and when combined with optical microscopy, different polymorphs in a mixture can quickly be spatially resolved. In this Application Note, the Edinburgh Instruments RM5 Raman Microscope is used to identify and discriminate between different polymorphs of the same drug compound.
Materials and Methods
Measurements were performed on an RM5 Raman Microscope equipped with a 532 nm laser, a 1200 gr/mm diffraction grating, and a back-illuminated CCD camera, Figure 1. Carbamazepine powder was purchased from Sigma Aldrich. The powder was heated at 170 ⁰C for 2 hours to induce a polymorphic phase transition.2

Figure 1. Edinburgh Instruments RM5 Raman Microscope with laser enclosure.
Polymorphism in Carbamazepine
The sample used for this work was carbamazepine (CBZ), or 5H-dibenzazepine-5-carboxamide, an anticonvulsant drug used in the treatment of epilepsy and as an adjunctive in treatments for schizophrenia and bipolar disorder. CBZ has five reported polymorphic structures, each with different dissolution rates and bioavailability.3 Commercial CBZ has a ρ-monoclinic crystalline structure, often referred to as CBZ III and is the most thermodynamically stable form at room temperature. Another polymorphic form is CBZ I, which has a triclinic crystallographic structure, Figure 2. CBZ III can be converted to CBZ I by heating it.
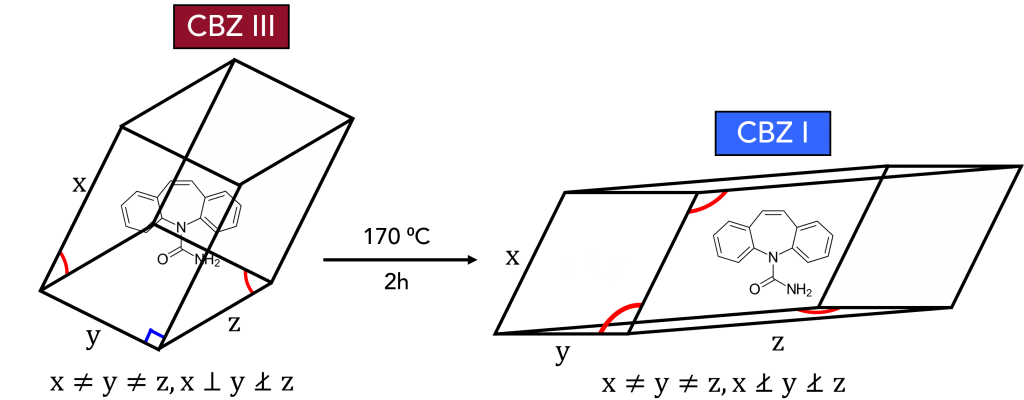
Figure 2. CBZ III to CBZ I polymorphic phase transition.
The two polymorphs were mixed and observed using optical darkfield imaging, Figure 3a. The two highlighted crystals in the image were analysed using Raman spectroscopy. Spectra were recorded at the areas indicated by the arrows on both crystals between 50 cm-1 and
3300 cm-1 and the spectra were segmented into three regions for easier visualisation of any subtle changes between the two polymorphs, Figure 3b-d.
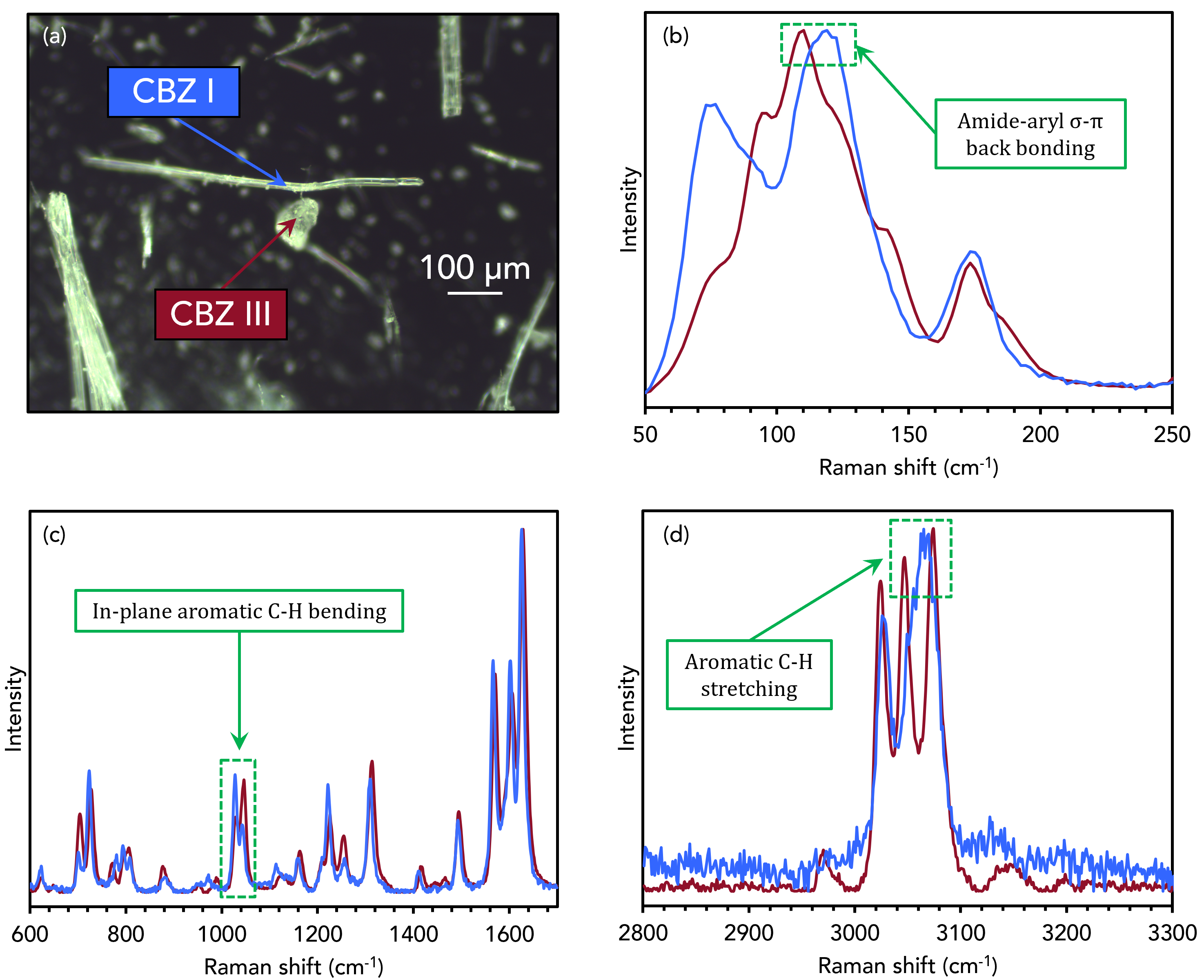
Figure 3. Raman analysis of CBZ III and CBZ I.
In the low wavenumber region in Figure 3b, which corresponds to intermolecular phonon modes in the crystal lattice, distinct spectral differences can be seen. The vibrational fingerprints in this region differ because the CBZ molecules adopt different packing orientations in the two crystal structures. The bands highlighted at 108 cm-1 and 120 cm-1 for CBZ III, and I are reported in the literature due to changes in σ-π back-bonding between the primary amide and aromatic groups during lattice vibrations.4
In Figure 3c, peak inversion between the two polymorphs at 1027 cm-1 and 1046 cm-1 can be assigned to in-plane bending vibrations of the aromatic C-H groups.5 Changes are also seen in Figure 3d between 3045 cm-1 and 3070 cm-1, corresponding to the stretching vibrations of the same C-H groups. The energies of these vibrations are altered when the packing orientation of the CBZ molecules changes during polymorphic phase transitions.
Conclusion
In this Application Note, Raman microscopy is a powerful method for discriminating pharmaceutical polymorphs. The high spectral resolution of the technique means that subtle changes in vibrational energies can be detected due to different packing orientations, and when combined with microscopy, the other crystalline structures can quickly be spatially resolved.
References
- D. Chistyakov et al., The Polymorphism of Drugs: New Approaches to the Synthesis of Nanostructured Polymorphs, Pharmaceutics, 2020, 12, 34.
- S. Fateixa et al., Carbamazepine polymorphism: A re-visitation using Raman imaging, Int. J. Pharm., 2022, 617, 121632.
- H. Yang et al., Selective crystallisation of carbamazepine polymorphs on surfaces with differing properties, CrystEngComm, 2017, 19, 6573.
- P. J. Larkin et al., Polymorph Characterization of Active Pharmaceutical Ingredients (APIs) Using Low-Frequency Raman Spectroscopy, Appl. Spectrosc., 2014, 68, 758-776.
- L. E. O’Brien et al., Use of in situ FT-Raman to study the kinetics of the transformation of carbamazepine polymorphs, J. Pharm., Biomed. Anal., 2004, 36, 335-340.









