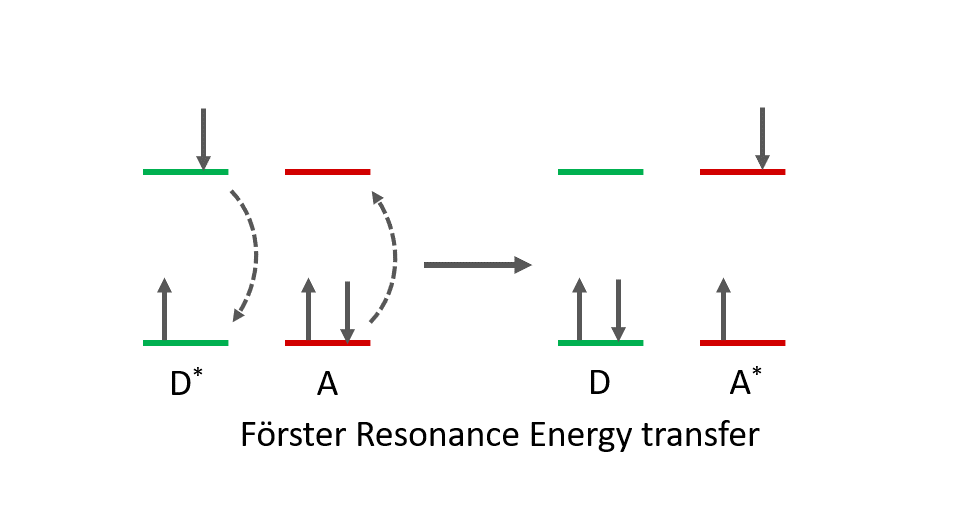
Förster resonance energy transfer (FRET) is a photophysical mechanism in which energy is transferred from an excited molecule (the donor) to another molecule (the acceptor) via a dipole-dipole resonance interaction.
When a donor molecule is photoexcited (D*), an oscillating dipole is generated and if the acceptor molecule (A) is in close proximity to the donor (typically between 1 and 10 nm), the dipole of the donor induces a dipole in the acceptor molecule, leading to the deactivation of the donor and the excitation of the acceptor (Figure 1).
Figure 1. FRET mechanism between a donor (D) and acceptor (A). Excited state molecules are represented by *.
There are two key requirements for FRET to occur:
Figure 2. Requirements for FRET.
The efficiency of energy transfer, η, is given by Eq. 1:
where r is the centre-to-centre distance between the donor and the acceptor and R0 is the Förster radius which is the distance at which the transfer efficiency is 50%. FRET is a dipole-dipole mechanism and the efficiency therefore drops off as 1 / r6. FRET will therefore only take place when the donor and acceptor are in close proximity, typically between 1 nm – 10 nm.
The Förster radius R0 is given by Eq. 2:
where k2 is the orientation factor and n is the refractive index of the medium containing the donor and acceptor. ΦD is the photoluminescence quantum yield of the donor in the absence of acceptor. J is the spectral overlap integral which quantifies the spectral overlap between the donor emission and acceptor absorption shown in Figure 2.
The other famous energy transfer is Dexter. An important difference between the two mechanisms is their dependence on distance (Figure 3). FRET is a dipole-dipole interaction mechanism which falls off as 1/r6 while Dexter is an electron exchange mechanism that requires orbital wavefunction orbital and the transfer efficiency therefore falls off much more rapidly (exponentially). FRET therefore occurs over longer distances than Dexter. The other important difference is that FRET is a singlet-only process while Dexter can proceed via both singlet and triplet excited states.
Figure 3: FRET and Dexter energy transfer dependence on distance. R0 is the Förster radius.